Incidental gall bladder findings, ranging from the more innocuous stones and mild CBD prominence to the more complex polyps, are common. ACR gave its recommendations on management/follow-up of incidental gall bladder and biliary findings in 2013 (you can read the closed-access article here).
This was followed by a more comprehensive guideline for managing incidental gall bladder polyps by the European Society of Gastrointestinal and Abdominal Radiology (ESGAR), in association with European Association for Endoscopic Surgery and other Interventional Techniques (EAES), International Society of Digestive Surgery – European Federation (EFISDS) and European Society of Gastrointestinal Endoscopy (ESGE) (you can read the open-access article here). In the absence of specific Indian guidelines, it is advisable to always follow the ESGAR guidelines whenever you come across a gall bladder polyp.
It is important to remember that all Indians are high-risk group for gall bladder cancer development as per the ESGAR guidelines, and the follow-up has to be accordingly performed irrespective of the patient’s age. Hence, for Indian patients (as well as for other high-risk patients such as those with age >50 years, those with sessile polyps, and with primary sclerosing cholangitis), the follow-up for gall bladder polyps essentially boils down to the chart below. The ACR guidelines can be followed for incidental gall bladder and biliary findings other than polyps (such as mild CBD prominence).
ESGAR Recommendations for Gall Bladder Polyp Management in High-Risk Patients

The overall ESGAR management algorithm is given below.
ESGAR Recommendations for Gall Bladder Polyp Management
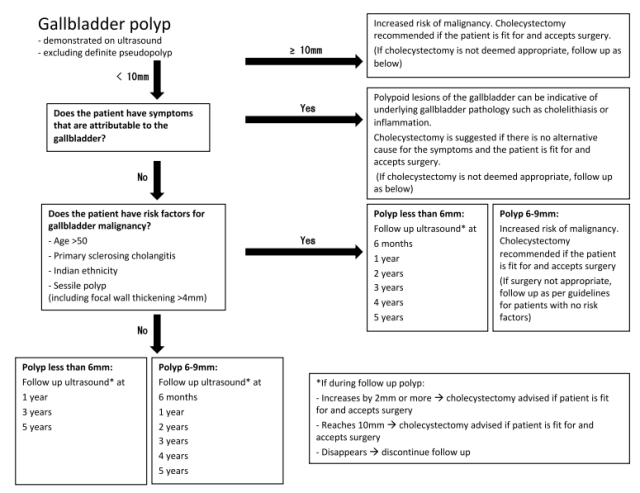
ACR Recommendations for Incidental findings of the Gall bladder and Bile ducts

– Akshay Baheti, Tata Memorial Hospital
PS: All images are a copyright of the original published article.

Pingback: Incidental Findings Follow-up Recommendations – Cafe Roentgen