A big thanks to Dr Govind Chavhan, Associate Pediatric Radiology Professor at the University of Toronto and the Hospital for Sick Children (Sick Kids), Toronto, for his his lovely talk. A summary of the key points in his talk as given below.
Utility of CT vs MRI for pediatric liver imaging:

MRI technique:
<5 yrs: sedation/anesthesia
>6-8 yrs: awake
Following sequences are generally taken:
- Coronal single shot T2
- Axial T2 TSE (fluid)
- Axial balanced SSFP (motion insensitive)
- Axial in and out phase (fat content of lesion, hemosiderosis)
- Axial diffusion (b: 50-100, lesion characterisation)
- Axial Thrive/VIBE pre
- Dynamic axial thrive/VIBE 4 runs (in smaller children, sequence run time is less, hence double NEX)
- Axial T1 TSE fs
- Axial thrive/VIBE 5 min delay (for hemangioma)
Dynamic imaging:
Pre-contrast: baseline for comparison, technical quality check, haemorrhage
Arterial: Contrast reached parenchyma, not sinusoids; any difference in arterial supply of parenchyma and lesion
Portal venous: vascular system / blood pool phase
Equilibrium phase: contrast similar in microvasculature and extracellular interstitial space
One may use hepatocyte specific contrast in select cases (e.g. evaluating possible FNH).
Multihance- 5% taken up by liver, hepatobiliary phase is 45min-1hr after injection
Primovist/Eovist- 50% taken up by liver, hepatobiliary phase is 20min after injection
When imaging with hepatocyte specific contrast, a few of the usual pre contrast sequences (such as T2W and DWI sequences) can be performed subsequent to contrast injection while waiting for the hepatobiliary phase.
PAEDIATRIC FOCAL LIVER LESIONS: Comprise 5-6% of paediatric abdominal masses.
BENIGN:
1. Hemangioma
- Most common
- Mostly infantile
- Large solitary/multiple small
- AFP normal
- No restriction of diffusion
- Can regress by 6-10yrs of age
- As time goes, they also become iso to liver on USG
- D/D: neuroblastoma metastases (liver will usually be enlarged in this though)
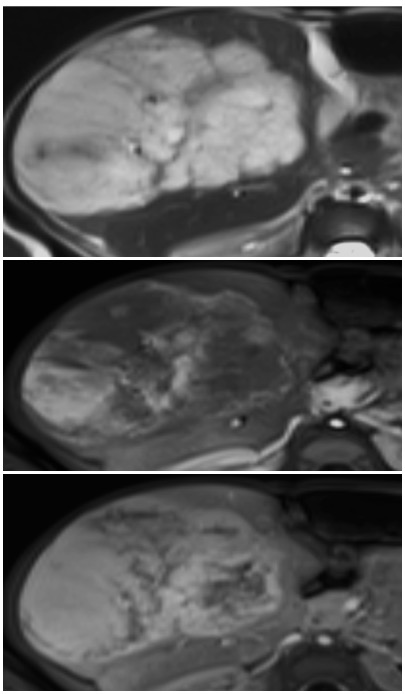
T2W, arterial and delayed post contrast images in a 3 month old demonstrate a T2 bright hepatic mass with peripheral arterial enhancement with delayed filling in, consistent with a hemangioma (infantile hemangioma is the technically correct term)
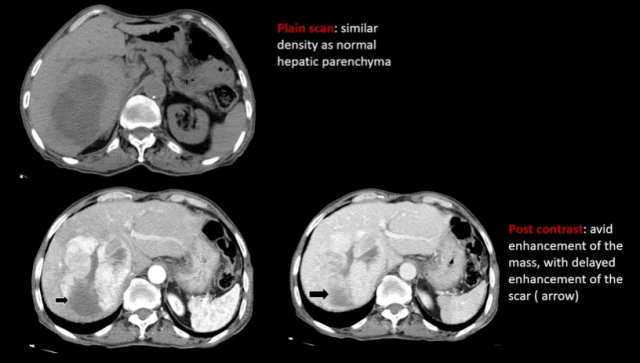
2. FNH
These can be seen de novo or in cancer survivors. Using hepatocyte specific contrast is a good way to diagnose FNH, as they will demonstrate uptake on the hepatobiliary phase images.
Healthy children Cancer survivors
-teenage girls -atypical features
-central T2 bright scar -smaller, multiple, no scar
-delayed enhancement of scar, -hyper on T2,prompt enhancement, then
-Rest shows prompt enhancement iso, intermediate restriction
with no washout
3. Regenerative nodules
- T2 dark
- T1 iso to hyper
- Children with Alagille syndrome often have large regenerative nodules
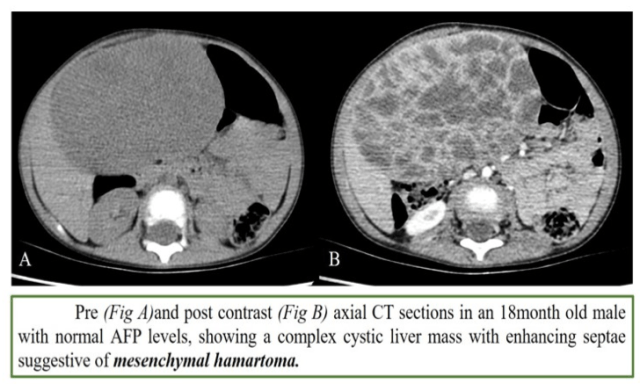
4. Mesenchymal hamartoma
- Neonates and infants
- Cystic
- AFP normal
5. Adenoma
- Rare
- Adolescents taking OC pills/ anabolic steroids
- Fat containing (<50% of cases)
- Haemorrhage
- Classified into 4 types, of which the inflammatory adenomas are the most common ones (>50%). HNFα mutated type commonly shows fat content. The beta catenin mutated type has a risk of malignant change and is the one to watch out for. The fourth type is the rare unclassified adenoma.

MALIGNANT
1. Hepatoblastoma
- 1-2% of all childhood cancers
- Peaks at <2yrs, and all occur before 4-5yrs
- 3rd most common after Wilms’ and neuroblastoma
- Malignant embryonal tumor
- Associated with prematurity
- AFP is elevated in 90% (normal AFP is seen in the sclerosing or small cell variety, which portends bad prognosis)
- Syndromes associated: Beckwith Weidman syndrome, fetal alcohol syndrome, etc. If one of the kidneys look enlarged or these is clinical hemihypertrophy, think of Beckwith Weidman syndrome. These kids need three-monthly surveillance on USG till 8 years of age to catch hepatoblastoma or Wilms tumor early.
- Types: These are most commonly either epithelial variety or mixed epithelial and mesenchymal on pathology. The mesenchymal component in the latter usually includes chondroid component which usually demonstrates calcification; hence a calcific liver mass in a child (particularly with raised AFP levels) is hepatoblastoma till proven otherwise.
- PRETEXT staging, a radiological staging classification, is used for baseline surgical evaluation in all cases. Please refer to the staging (google it!) and report it in every case of hepatoblastoma. You can find a brief outline here: https://radiopaedia.org/articles/paediatric-liver-tumour-pretext-grouping-system
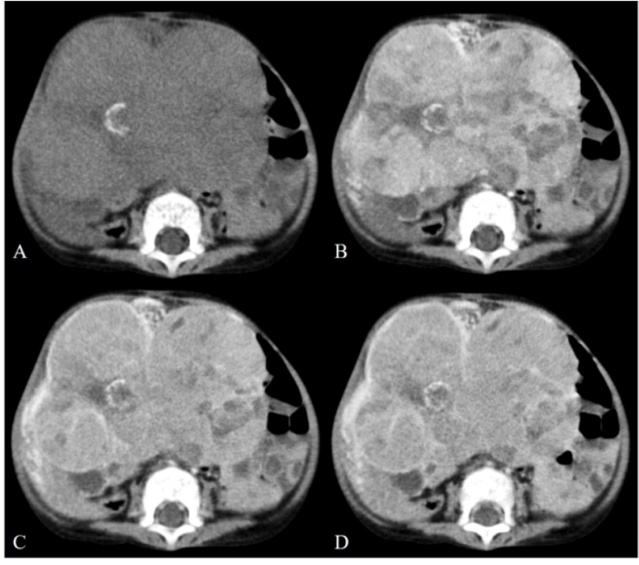
3 year old boy with a large heterogeneous hepatic mass with calcifications, consistent with PRETEXT IV hepatoblastoma.
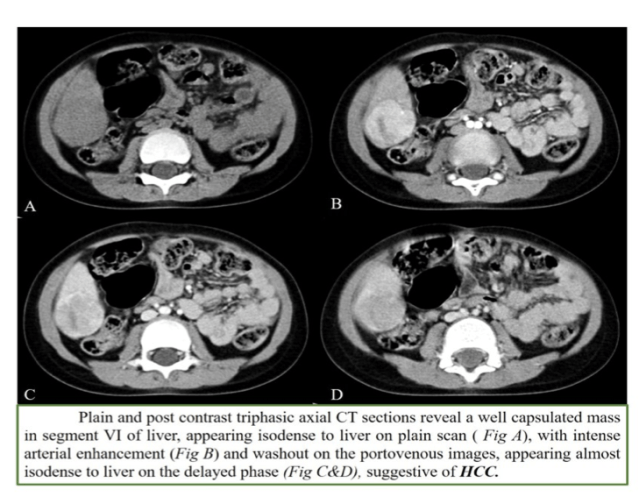
2. HCC
- Age: 10-14yrs
- With or without cirrhosis
- AFP can be elevated in 70% of cases
- Arterial phase enhancement with early washout (especially in cirrhosis-tyrosinemia etc)
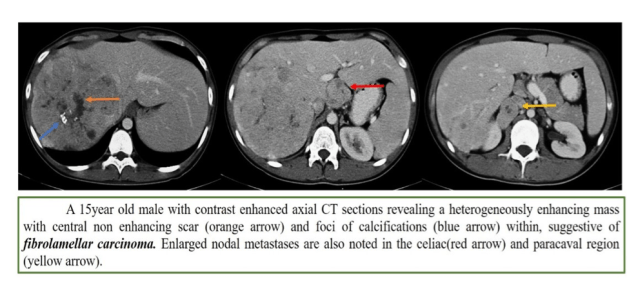
3. Fibrolamellar HCC
- Better prognosis
- Liver lesion looking like spleen on all sequences is bad
- Scar is dark on all sequences (unlike FNH, where it is bright on T2WI)
4. Undifferentiated embryonal sarcoma
- 6-10 years
- AFP is normal
- Solid-cystic; the mass usually has a large cystic/necrotic component which is quite classic in this age group for this entity
- D/D: mesenchymal hamartoma (happens at a younger age)

7 year old boy with an almost cystic appearing hepatic mass with thick peripheral enhancement. In the absence of fever, think of embryonal sarcoma as a possible diagnosis rather than an abscess; as this was correctly called.
Most useful factors for differentiation:
- Age
- AFP
- MRI

DIFFUSE LIVER DISEASES:
1. Hepatic steatosis
- NAFLD (non alcoholic fatty liver disease)
- If inflammatory: NASH (non alcoholic steatohepatitis). The predisposing factors for NAFLD and NASH include: obesity, insulin resistance, hypertriglyceridemia, drug hepatotoxicity, genetic and metabolic factors. A few of the patients with NAFLD progress to NASH. Similarly, patients with NASH can progress to cirrhosis. It hence needs evaluation and treatment.
- Liver biopsy may be performed in select cases.
- Quantification and degree of fatty change: MRI and MR spectroscopy (most accurate) can be used.
- Normal liver fat : <5%
2. Fibrosis and cirrhosis
- Fibrosis: excess deposition of collagen, proteoglycans and other macromolecules
- Cirrhosis: formation of nodules of regenerative hepatocytes surrounded by fibrous bands
- Causes: biliary atresia, α1 antitrypsin deficiency, PSC, autoimmune hepatitis, hemochromatosis, hep B and C (rare), Wilsons’, Alagille, cystic fibrosis, primary biliary cirrhosis
- Morphologic features like right lobe atrophy, left lobe and caudate lobe hypertrophy, surface nodularity, presence of regenerative nodules, fibrous septa, or heterogeneous enhancement on dynamic imaging have good specificity but poor sensitivity for identifying cirrhosis. If they are present they are diagnostic of fibrosis/cirrhosis, but if they are absent it does not exclude fibrosis/cirrhosis.
- Regenerative nodules without cirrhosis are seen in NRH, portal vein thrombosis, and hepatic necrosis
- Significant fibrosis without regenerative nodules seen in congenital hep fibrosis
- Surface nodularity can be seen in fulminant hepatic failure without cirrhosis
- MR elastography: modality of choice for quantifying fibrosis; can replace biopsy
3. Iron overload
- Hemochromatosis, defects in heme metabolism, long term transfusion
- MR estimates iron overload qualitatively or quantitatively. Qualitative sequences include the in and out phase sequences, in which the iron loaded liver parenchyma becomes darker on the in phase sequence. Quantitative sequences use increasing TE (echo time). With increasing TE, the iron overloaded parenchyma turns more and more dark.
If anybody is interested in liver imaging in more details (even for adults), this Radiographics article by Dr Chavhan would be extremely useful for understanding concepts better.
– Nikshita Jain, Senior registrar, Tata Memorial Hospital
– Akshay Baheti, Assistant Professor, Tata Memorial Hospital

Thanks a lot for sharing
LikeLike